Table of Contents
Orally dissolving medications that contains buprenorphine are joined to serious dental challenges, such as complete tooth reduction, the US Food items and Drug Administration (Fda) warns in a protection interaction.
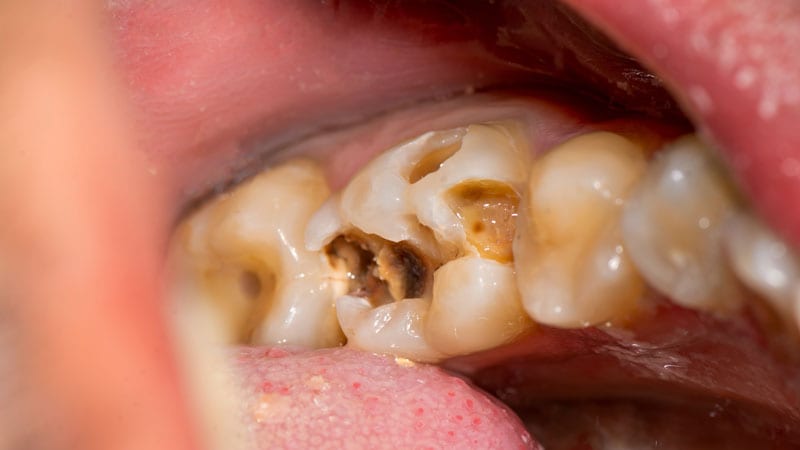
The oral side outcomes of these medications, which are used to handle opioid use condition (OUD) and discomfort, consist of cavities/tooth decay, including rampant caries dental abscesses/an infection tooth erosion fillings slipping out and, in some circumstances, total tooth loss.
A number of cases have been claimed even in individuals with no record of dental problems.
The Fda is including a warning about the risk of dental problems to the prescribing information and the individual treatment guidebook for all buprenorphine-containing medicines dissolved in the mouth.
The Food and drug administration emphasizes, even so, that buprenorphine remains “an important therapy possibility for OUD and suffering, and the gains of these medications clearly outweigh the risks.”
Much more Than 300 Described Conditions
Buprenorphine was permitted in 2002 as a sublingual tablet, and in 2015 as a film to be put within the cheek to handle soreness. Both equally shipping techniques have been involved with dental issues.
Since buprenorphine was permitted, the Fda has discovered 305 cases of dental challenges linked with orally dissolving buprenorphine, which include 131 classified as critical.
There may possibly be other instances, the Fda states, as these signifies only situations documented to the Fda or released in the clinical literature.
The average age of the people who made dental complications even though taking buprenorphine is 42 many years, but these as young as 18 many years previous have been also afflicted.
Most cases occurred in patients working with the medicines for OUD however, 28 conditions of dental difficulties happened in clients utilizing it to take care of agony.
In 26 instances, people experienced no prior heritage of dental issues. Some dental challenges formulated as quickly as 2 months following procedure commenced the median time to analysis was about 2 a long time just after setting up cure.
Among the all 305 conditions reported, 113 included two or much more tooth.
The most common treatment for the dental challenges was tooth extraction/elimination, which was reported in 71 instances. Other scenarios expected root canals, dental surgical treatment, and other procedures this kind of as crowns and implants.
Recommendations
The Fda suggests health care suppliers should really counsel individuals that serious and considerable tooth decay, tooth reduction, and tooth fracture have been documented with the use of transmucosal buprenorphine-that contains medications and emphasize the relevance of checking out their dentist to closely keep an eye on their enamel.
People ought to be recommended to carry on taking buprenorphine remedies as prescribed and not stop instantly devoid of first chatting to their health care company as this could direct to major consequences, which includes relapse, misuse or abuse of other opioids, overdose, and demise.
Clients are also getting recommended to acquire added methods to support reduce the hazard of severe dental complications.
People should really also be educated on approaches to retain or boost oral health although using transmucosal buprenorphine medications.
Following the medicine is absolutely dissolved, the individual need to acquire a massive sip of drinking water, swish it carefully all around the enamel and gums, swallow, and wait at minimum 1 hour just before brushing their teeth, the Food and drug administration advises. This will let time for the mouth to step by step return to oral homeostasis and avoid any mechanical damage that may well occur thanks to brushing.
The Food and drug administration also advises that sufferers inform their service provider about any background of tooth challenges, such as cavities, and schedule a dentist check out before long right after starting up the medicine.
Dental difficulties relevant to transmucosal buprenorphine-containing medicines need to be claimed to the FDA’s MedWatch program.
For far more Medscape Psychiatry news, sign up for us on Twitter and Fb.




More Stories
Do you need a dentist visit every 6 months? That filling? The data is weak
FDA said it “never” inspected dental lab that made controversial AGGA device
Prolonged hold out-situations and not sufficient dentists: Rollout of new dental program far from painless